Retargeting Tips For Highly-Regulated Advertisers
“Should or should I not include retargeting in a media plan?” is a regular question asked by clients. Although it is a fantastic technology and when used properly can drive huge results — clients are often oversold and promised the world — and it is ever so difficult for them to cut through the clutter. […]
“Should or should I not include retargeting in a media plan?” is a regular question asked by clients. Although it is a fantastic technology and when used properly can drive huge results — clients are often oversold and promised the world — and it is ever so difficult for them to cut through the clutter.
For direct-to-consumer advertisers and agencies, retargeting may be a no brainer. But for more regulated companies — such as those in healthcare and finance verticals — the questions are much more complex and not as easily answered.
A Quick Recap On Retargeting
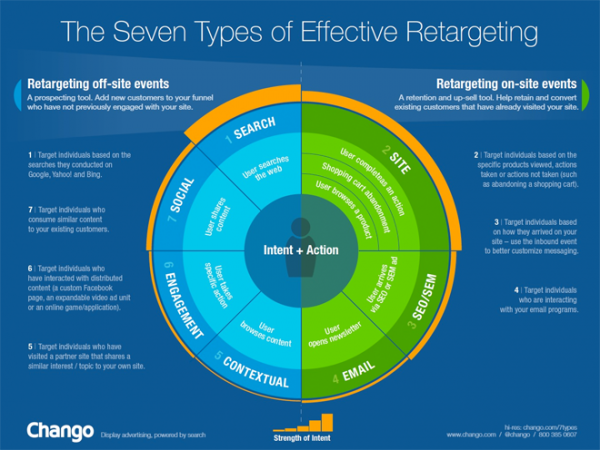
There are two types of retargeting. One is where we target ‘off-site’ events – things that potential customers are doing off of your website; and the other where we target ‘on-site’ events – things that your customers are doing on your website. A combination of both of these methods will bring the highest performance.
Off-Site Retargeting
For off-site retargeting, search retargeting is the most common form. Using cookies already on the individual’s computer, search-retargeting looks at the keywords an individual is searching for, matches those keywords to our display campaign, and allows us to show them a display ad outside of the search engine. This is a strong form of retargeting as the individual is showing intent for the brand, by what they are searching for.
On-Site Retargeting
For on-site retargeting, site retargeting is the most common form. Site retargeting allows us to retarget individuals that have visited our website, by placing or activating a cookie on their computer. Here, we know that the individual is interested in our brand because they have been to our website. Our ads can be tailored to either provide them with additional information or make them come back for more.
There Is Some Regulation Such As Opt-Out
As an example, today there is no firm regulation from the FDA on whether or not you can retarget users – specifically in the pharmaceutical and medical fields. However, companies are urged to not overuse this technology and to be mindful of what could be considered “spam.” Consumers are asked to take more responsibility to disable or enable tracking of themselves.
Anonymous Retargeting
Reputable ad networks and publishers are retargeting users completely anonymously. No personal identifiable information (PII) is shared between the user and the publisher, even if the brand is retargeting visitors to their own website. Publishers, such as Google, go as far as to mandate that no PII is sent to them via their Privacy Policy.
AdChoices
The White House, Department of Commerce and the Federal Trade Commission have all commended the Digital Advertising Alliance and its partners for the significant progress toward consumer privacy with their Self Regulatory Program. A product of this program, AdChoices, is now an industry standard on all ads that are being behaviorally targeted and allow consumers to opt out of this type of targeting.
A Couple Thoughts After You Decide To Use Retargeting
Retargeting is a very powerful advertising program that brands should take advantage of. However, this should not be the only program they are relying on, and the program should be developed in good taste.
Use AdChoices
Although it is a best practice of all of the major ad networks, it is a good idea to validate with the ad network that their ads will include the AdChoices icon allowing consumers to opt out of behavioral targeting.
Cap Interactions
When setting up your retargeting campaign, some recommend the 7×7 rule: limit the window to 7 days or less (users who have been to your site within the last 7 days can see a retargeting ad), and cap the impressions to 7 per person per day. This will limit the exposure of a retargeted ad to any specific user — and limit the ‘stalker factor’ of the brand.
Update The Site’s Privacy Policy
Although this is not required, it is suggested to update the site’s privacy policy to include display advertising and retargeting. Google recommends that the privacy policy be updated whenever Google Analytics is used, and goes further to recommend additional updates when analytics is being used for display advertising and retargeting. Take a look at the FDA’s own privacy policy here.
Bottom line: retargeting should be used on a case-by-case basis — and if a client is overly hesitant, the risk may outweigh the reward. Although advertisers can use retargeting to drive highly relevant ads to their potential audience with high ROI — they must also be aware of increasing consumer privacy awareness and ever-changing regulatory factors.
Contributing authors are invited to create content for Search Engine Land and are chosen for their expertise and contribution to the search community. Our contributors work under the oversight of the editorial staff and contributions are checked for quality and relevance to our readers. The opinions they express are their own.
Related stories
New on Search Engine Land